3 results
Factors associated with SARS-CoV-2 and community-onset invasive Staphylococcus aureus coinfection, 2020
- Kelly Jackson, Sydney Resler, Joelle Nadle, Susan Petit, Susan Ray, Lee Harrison, Ruth Lynfield, Kathryn Como-Sabetti, Carmen Bernu, Ghinwa Dumyati, Marissa Tracy, William Schaffner, Holly Biggs, Isaac See
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 3 / Issue S2 / June 2023
- Published online by Cambridge University Press:
- 29 September 2023, pp. s84-s85
-
- Article
-
- You have access Access
- Open access
- Export citation
-
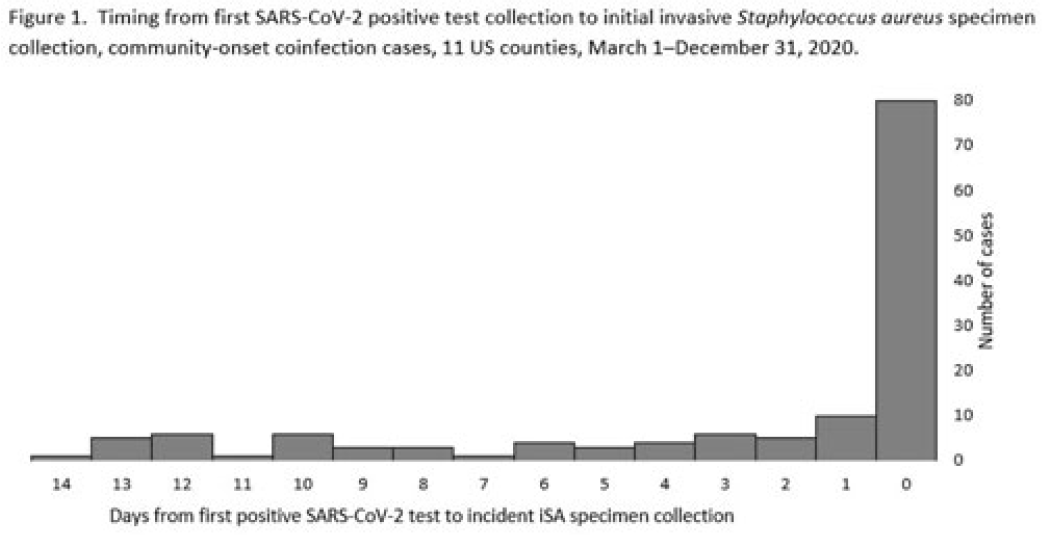
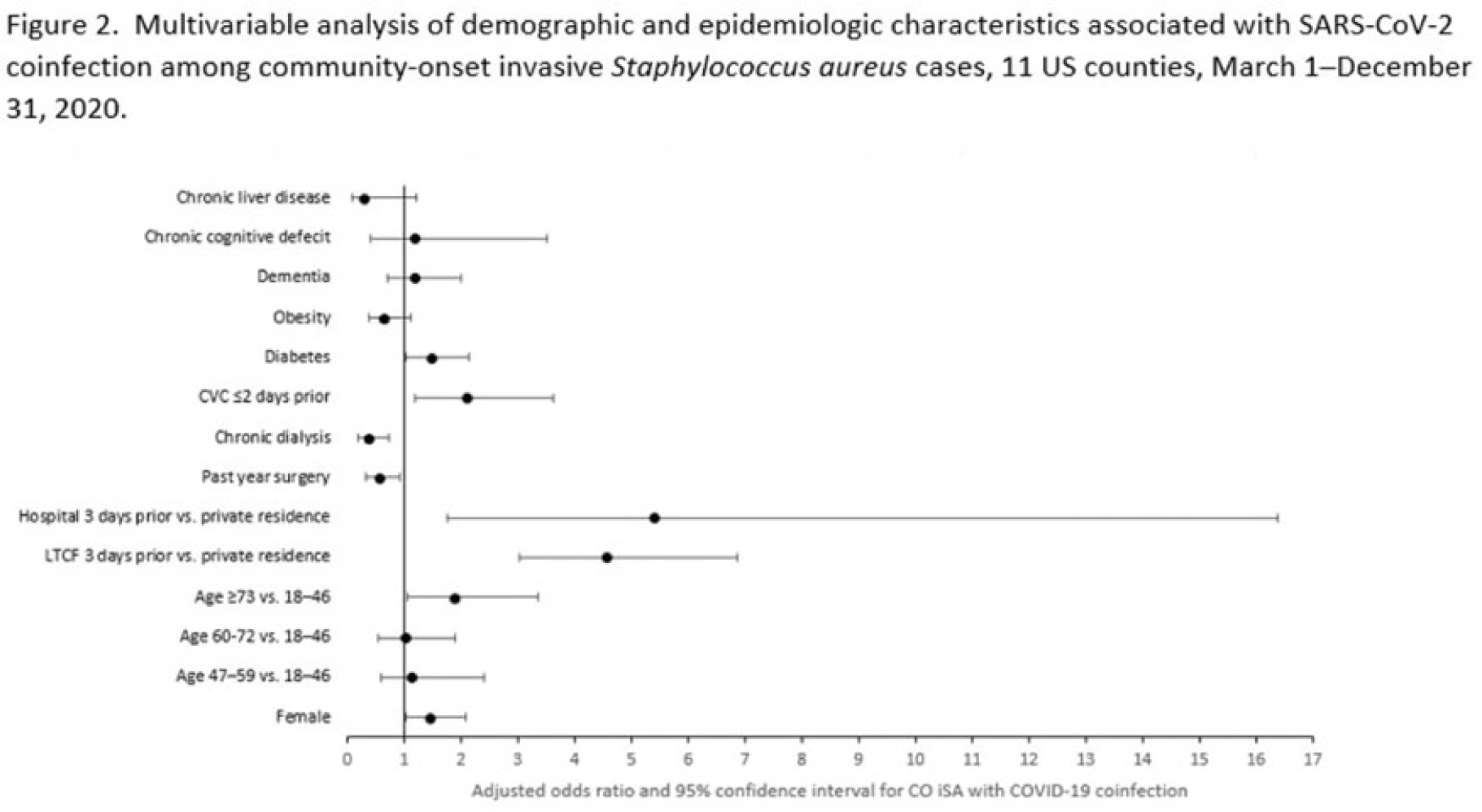
Background: Previous analyses describing the relationship between SARS-CoV-2 infection and Staphylococcus aureus have focused on hospital-onset S. aureus infections occurring during COVID-19 hospitalizations. Because most invasive S. aureus (iSA) infections are community-onset (CO), we characterized CO iSA cases with a recent positive SARS-CoV-2 test (coinfection). Methods: We analyzed CDC Emerging Infections Program active, population- and laboratory-based iSA surveillance data among adults during March 1–December 31, 2020, from 11 counties in 7 states. The iSA cases (S. aureus isolation from a normally sterile site in a surveillance area resident) were considered CO if culture was obtained <3 days after hospital admission. Coinfection was defined as first positive SARS-CoV-2 test ≤14 days before the initial iSA culture. We explored factors independently associated with SARS-CoV-2 coinfection versus no prior positive SARS-CoV-2 test among CO iSA cases through a multivariable logistic regression model (using demographic, healthcare exposure, and underlying condition variables with P<0.25 in univariate analysis) and examined differences in outcomes through descriptive analysis. Results: Overall, 3,908 CO iSA cases were reported, including 138 SARS-CoV-2 coinfections (3.5%); 58.0% of coinfections had iSA culture and the first positive SARS-CoV-2 test on the same day (Fig. 1). In univariate analysis, neither methicillin resistance (44.2% with coinfection vs 36.5% without; P = .06) nor race and ethnicity differed significantly between iSA cases with and without SARS-CoV-2 coinfection (P = .93 for any association between race and ethnicity and coinfection), although iSA cases with coinfection were older (median age, 72 vs 60 years , P<0.01) and more often female (46.7% vs 36.3%, P=0.01). In multivariable analysis, significant associations with SARS-CoV-2 coinfection included older age, female sex, previous location in a long-term care facility (LTCF) or hospital, presence of a central venous catheter (CVC), and diabetes (Figure 2). Two-thirds of co-infection cases had ≥1 of the following characteristics: age > 73 years, LTCF residence 3 days before iSA culture, and/or CVC present any time during the 2 days before iSA culture. More often, iSA cases with SARS-CoV-2 coinfection were admitted to the intensive care unit ≤2 days after iSA culture (37.7% vs 23.3%, P<0.01) and died (33.3% vs 11.3%, P<0.01). Conclusions: CO iSA patients with SARS-CoV-2 coinfection represent a small proportion of CO iSA cases and mostly involve a limited number of factors related to likelihood of acquiring SARS-CoV-2 and iSA. Although CO iSA patients with SARS-CoV-2 coinfection had more severe outcomes, additional research is needed to understand how much of this difference is related to differences in patient characteristics.
Disclosures: None
Increases in methicillin-sensitive Staphylococcus aureus bloodstream infection incidence, 2016–2019
- Kelly Jackson, Joelle Nadle, Susan Ray, Ruth Lynfield, Ghinwa Dumyati, Marissa Tracy, William Schaffner, David Ham, Isaac See
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 2 / Issue S1 / July 2022
- Published online by Cambridge University Press:
- 16 May 2022, pp. s63-s64
-
- Article
-
- You have access Access
- Open access
- Export citation
-
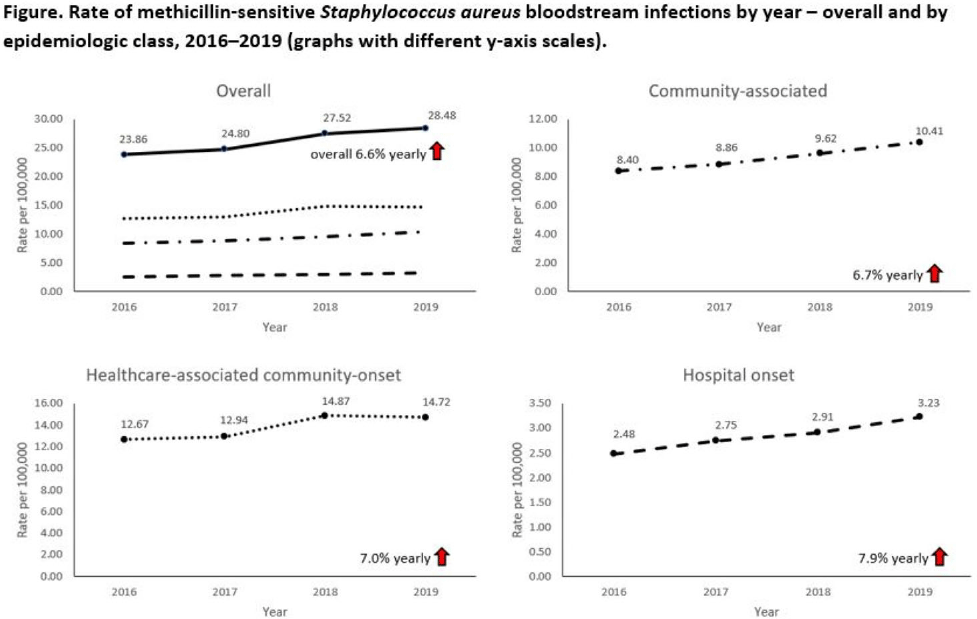
Background: Incidence of methicillin-sensitive Staphylococcus aureus (MSSA) bloodstream infections (BSIs) in the United States during 2012–2017 has been reported to have been stable for hospital-onset BSIs and to have increased 3.9% per year for community-onset BSIs. We sought to determine whether these trends continued in more recent years and whether there were further differences within subgroups of community-onset BSIs. Methods: We analyzed CDC Emerging Infections Program active, population- and laboratory-based surveillance data during 2016–2019 for MSSA BSIs from 8 counties in 5 states. BSI cases were defined as isolation of MSSA from blood in a surveillance area resident. Cases were considered hospital onset (HO) if culture was obtained >3 days after hospital admission and healthcare-associated community-onset (HACO) if culture was obtained on or after day 3 of hospitalization and was associated with dialysis, hospitalization, surgery, or long-term care facility residence within 1 year prior or if a central venous catheter was present ≤2 days prior. Cases were otherwise considered community-associated (CA). Annual rates per 100,000 census population were calculated for each epidemiologic classification; rates of HACO cases among chronic dialysis patients per 100,000 dialysis patients were calculated using US Renal Data System data. Annual increases were modeled using negative binomial or Poisson regression and accounting for changes in the overall population age group, and sex. Descriptive analyses were performed. Results: Overall, 8,344 MSSA BSI cases were reported. From 2016–2019 total MSSA BSI rates increased from 23.9 per 100,000 to 28.5 per 100,000 (6.6% per year; P < .01). MSSA BSI rates also increased significantly among all epidemiologic classes. HO cases increased from 2.5 per 100,000 to 3.2 per 100,000 (7.9% per year; P = .01). HACO cases increased from 12.7 per 100,000 to 14.7 per 100,000 (7.0% per year; P = .01). CA cases increased from 8.4 per 100,000 to 10.4 per 100,000 (6.7% per year; P < .01) (Fig. 1). Significant increases in MSSA BSI rates were also observed for nondialysis HACO cases (9.3 per 100,000 to 11.1 per 100,000; 7.8% per year; P < .01) but not dialysis HACO cases (1,823.2 per 100,000 to 1,857.4 per 100,000; 1.4% per year; P = .59). Healthcare risk factors for HACO cases were hospitalization in the previous year (82%), surgery (31%), dialysis (27%), and long-term care facility residence (19%). Conclusions: MSSA BSI rates increased from 2016–2019 overall, among all epidemiologic classes, and among nondialysis HACO cases. Efforts to prevent MSSA BSIs among individuals with healthcare risk factors, particularly those related to hospitalization, might have an impact on MSSA BSI rates.
Funding: None
Disclosures: None
Trends in Staphylococcus aureus Bloodstream Infections in Nursing Homes in Monroe County, New York
- Marissa Tracy, Christina B. Felsen, Anita Gellert, Ghinwa Dumyati
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 41 / Issue S1 / October 2020
- Published online by Cambridge University Press:
- 02 November 2020, pp. s417-s418
- Print publication:
- October 2020
-
- Article
-
- You have access Access
- Export citation
-
Background: Methicillin-resistant Staphylococcus aureus (MRSA) bloodstream infections (BSIs) are common in hospitals and nursing homes. Infection prevention efforts reduced MRSA BSI in hospitals but the trend in nursing homes is not well described. In addition, the contribution of methicillin sensitive S. aureus (MSSA) to the total burden of invasive S. aureus (iSA) in nursing homes remains unknown. Methods: As part of the CDC Emerging Infections Program, we conduct population-based surveillance for iSA infections in Monroe County, New York. Case patients were county residents with S. aureus isolated from a sterile site. Our analysis was limited to data from 2009–2018 for MRSA and 2015–2018 for MSSA and to cases classified as hospital-onset (HO, positive culture ≥3 calendar days after admission) or nursing home-onset (NHO, positive culture in nursing homes or within 3 days of hospital admission from a nursing home). Risk factors for iSA BSI in nursing homes were compared using the χ2 and Student t tests in SAS version 9.4 software. Results: During 2009–2014, 664 MRSA cases occurred and 427 (64%) were BSIs. Of these, 228 (53%) were NHO and 199 (47%) were HO. The BSI incidence per 100,000 population of NHO cases declined from 7.9 in 2009 to 2.8 in 2014, mirroring the decline in HO incidence from 8.7 in 2009 to 3.1 in 2014 (Fig. 1). During 2015–2018, 203 MRSA cases (163 BSIs, 80%) and 235 MSSA cases (163 BSIs, 69%) occurred. Of the 163 MRSA BSIs, 94 (58%) were NHO and 69 (42%) were HO, whereas of the 235 MSSA BSIs, only 56 (34%) were NHO and 107 (66%) were HO. MRSA BSI incidence per 100,000 population in both settings plateaued during 2015–2018 (Fig. 1) and MSSA NHO BSI incidence was lower than HO (1.9 NHO vs 3.6 HO). The total iSA BSI incidence was similar in both settings (5.9 vs 5.0 per 100,000 population in HO and NHO, respectively). NHO MSSA and MRSA cases have similar risk factors for BSI; 45 (30%) had decubitus ulcers, 34 (23%) were on chronic dialysis, 41 (27%) had a CVC in place within 2 days of BSI onset, and 63% had prior healthcare exposures. Most of these developed within 4 weeks of hospital discharge (Fig. 2). Conclusions: The incidence of MRSA BSI in nursing homes has declined since 2009 but plateaued starting in 2015. Compared to MRSA, MSSA caused fewer BSIs in nursing homes; however, iSA risk factors, including previous healthcare exposure, were similar. Continued study is needed to identify interventions effective against all iSA infections in nursing homes.
Funding: None
Disclosures: None






